|
Slater JC (1964) Journal of Chemical Physics 39:3199- Crystal Radii The bond length between atoms A and B is the sum of the atomic radii,ĬrystalMaker uses Atomic-Ionic radii data from: For example, the atomic-ionic radius of chlorine (Cl -) is larger than its atomic radius. 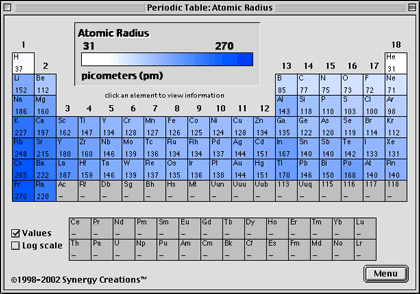
These are the "realistic" radii of atoms, measured from bond lengths in real crystals and molecules, and taking into account the fact that some atoms will be electrically charged. CrystalMaker uses Van-der-Waals Radii data from:īondi A (1964) Journal of Physical Chemistry 68:441- Atomic-Ionic Radii Van-der-Waals radii are determined from the contact distances between unbonded atoms in touching molecules or atoms. Whilst this is straightforward for some molecules such as Cl 2 and O 2, in other cases one has to infer the covalent radius by measuring bond distances to atoms whose radii are already known (e.g., a C-X bond, in which the radius of C is known).ĬrystalMaker uses covalent radii listed on CrystalMaker-user Mark Winter's excellent Web Elements website. The covalent radius of an atom can be determined by measuring bond lengths between pairs of covalently-bonded atoms: if the two atoms are of the same kind, then the covalent radius is simply one half of the bond length. Springer Verlag, Berlin.Ĭlementi E, Raimondi DL, Reinhardt WP (1963). Vainshtein BK, Fridkin VM, Indenbom VL (1995) Structure of Crystals (3rd Edition). CrystalMaker uses Atomic radii data from two sources: Although more electrons are being added to atoms, they are at similar distances to the nucleus and the increasing nuclear charge "pulls" the electron clouds inwards, making the atomic radii smaller.Ītomic radii are generally calculated, using self-consistent field functions. Atomic radii decrease, however, as one moves from left to right, across the Periodic Table. The general trend is that atomic sizes increase as one moves downwards in the Periodic Table of the Elements, as electrons fill outer electron shells. Atomic RadiiĪtomic radii represent the sizes of isolated, electrically-neutral atoms, unaffected by bonding topologies. In the following article we'll explore a number of different sets of distinct atomic radius sizes, and later we'll see how you can make use of these "preset" values with CrystalMaker. Take the humble carbon atom as an example: in most organic molecules a covalently-bonded carbon atom is around 1.5 Ångstroms in diameter (1 Ångstrom unit = 0.1 nanometres = 10 -10 metres) but the same atom in an ionic crystal appears much smaller: around 0.6 Ångstroms. However, even for atoms of the same type, atomic radii can differ, depending on the oxidation state, the type of bonding and - especially important in crystals - the local coordination environment. To a first approximation we can regard atoms as "hard spheres", with an outer radius defined by the outer electron orbitals. Therefore, there are various non-equivalent definitions of atomic radius.How big is an atom? A simple question maybe, but the answer is not at all straighforward. However, this assumes the atom to exhibit a spherical shape, which is only obeyed for atoms in vacuum or free space. 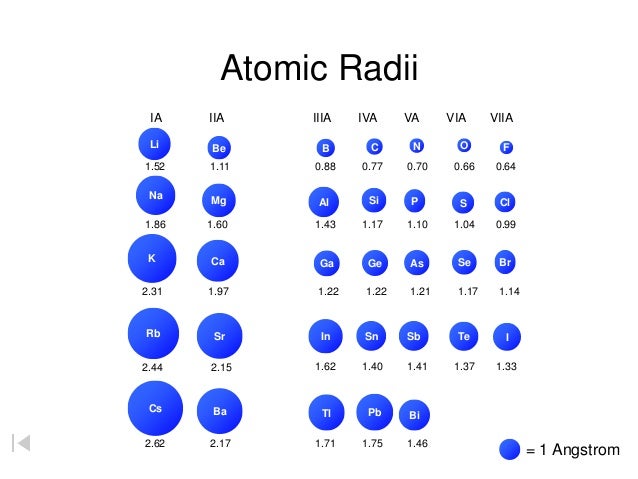
The atomic radius of a chemical element is a measure of the distance out to which the electron cloud extends from the nucleus. 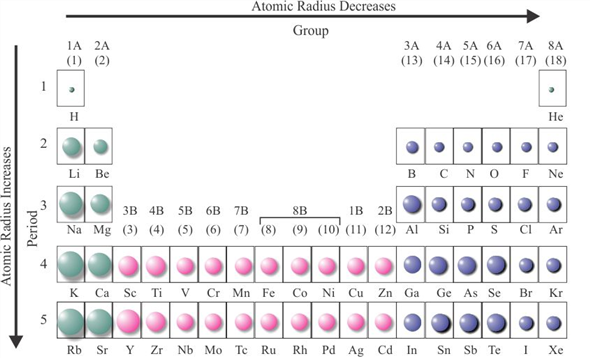
It must be noted, atoms lack a well-defined outer boundary. The atomic radius of Francium atom is 260pm (covalent radius). Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally-occuring isotopes and their abundance. The atomic mass is carried by the atomic nucleus, which occupies only about 10 -12 of the total volume of the atom or less, but it contains all the positive charge and at least 99.95% of the total mass of the atom. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Mass numbers of typical isotopes of Francium are 223. Isotopes are nuclides that have the same atomic number and are therefore the same element, but differ in the number of neutrons. The difference between the neutron number and the atomic number is known as the neutron excess: D = N – Z = A – 2Z.įor stable elements, there is usually a variety of stable isotopes. Neutron number plus atomic number equals atomic mass number: N+Z=A. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol N. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10 -19 coulombs. 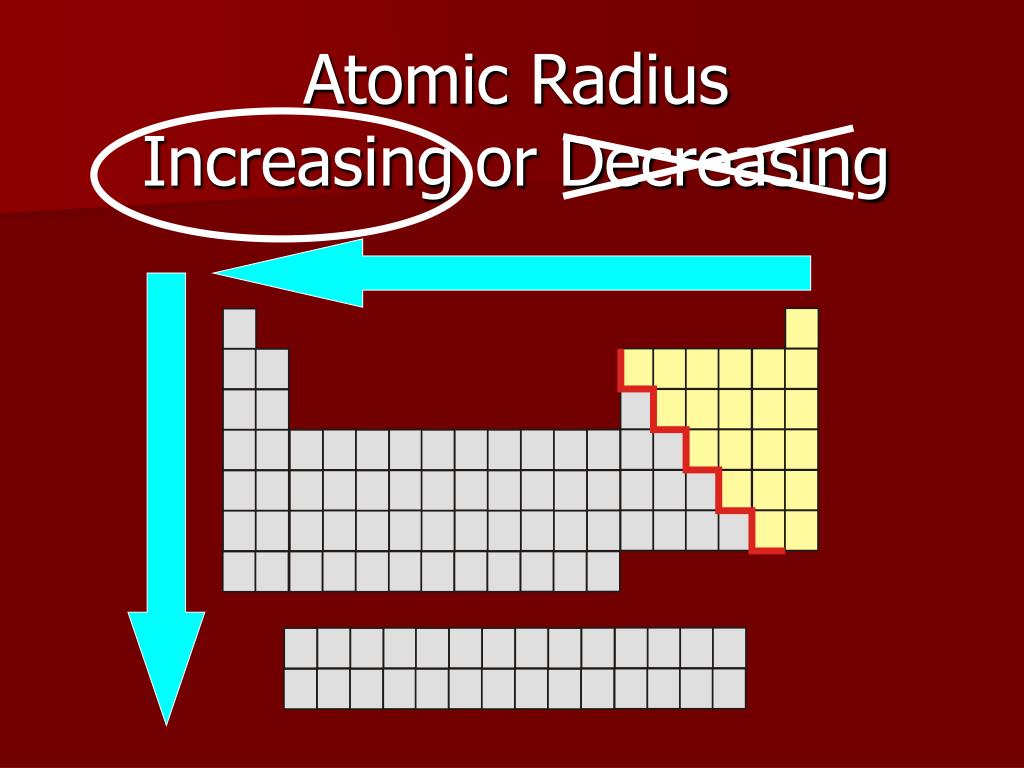
Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. Atomic Number – Protons, Electrons and Neutrons in Franciumįrancium is a chemical element with atomic number 87 which means there are 87 protons in its nucleus.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
